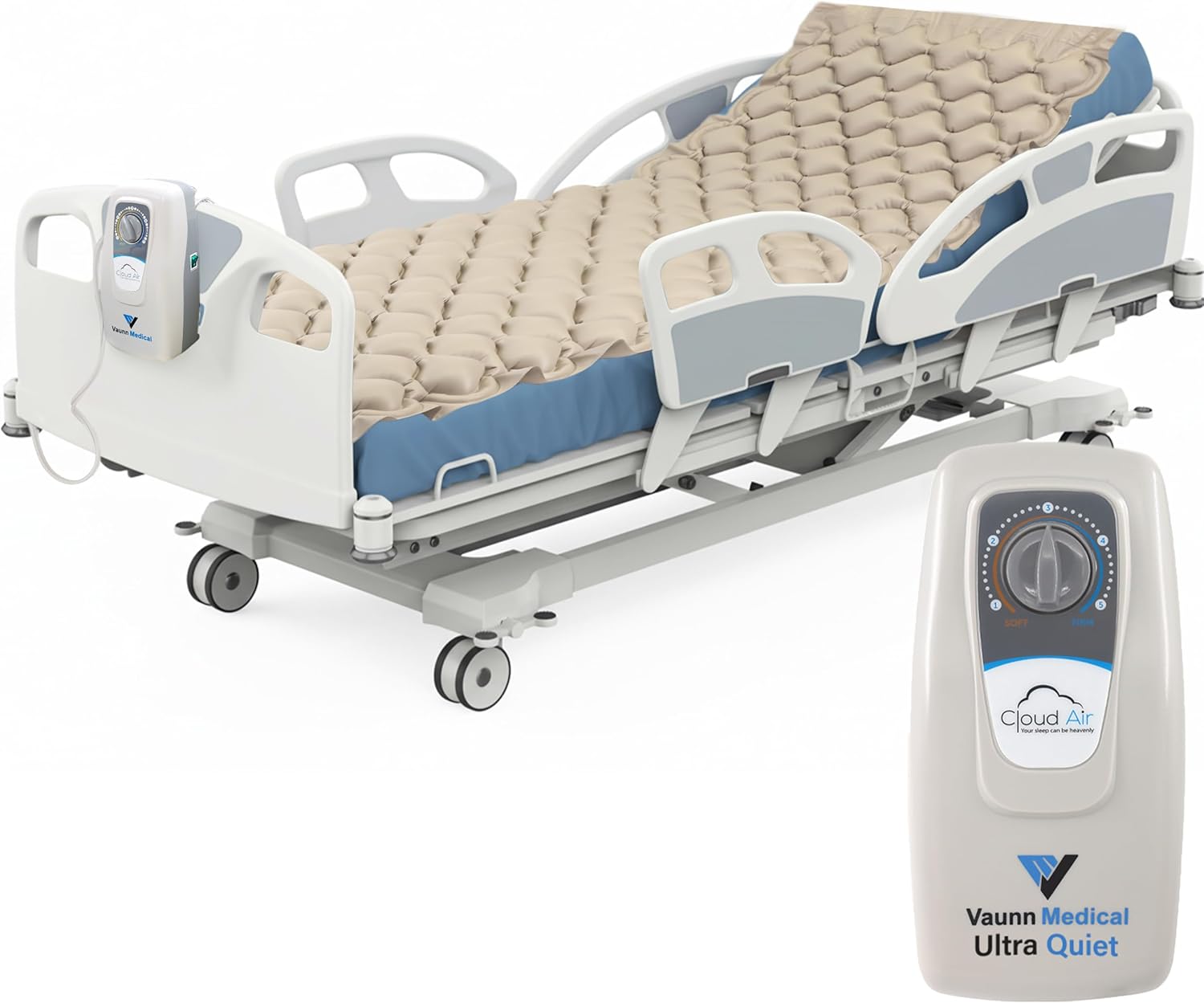
Drive Medical 14001E Med-Aire Alternating Pressure Pump and Pad System, 78" x 36" (Variable Pressure Pump)
$75.25
Price when purchased online
Free shipping
Free 30-day returns
Sold and shipped by el42.de
We aim to show you accurate product information. Manufacturers, suppliers and others provide what you see here.
Product details
| Management number | 211735911 | Release Date | 2026/04/04 | List Price | $30.10 | Model Number | 211735911 | ||
|---|---|---|---|---|---|---|---|---|---|
| Category | |||||||||
Drive Medical's Med Aire Alternating Pressure Pump and Pad System with low air loss provides while alternating pressure and is designed to be used in the prevention, treatment and management of pressure ulcers. The bubble pad provides superior therapeutic treatment alternating which of the 130 individual bubble cells are inflated or deflated every 5 minutes, never placing too much pressure on any affected area. The variable pressure 4 LPM (liters per minute) pump produces a consistent airflow, adjustable pressure, and its quiet technology alternately inflates and deflates the pad's air cells.
- Vinyl
- Alternating Pressure Air Mattress Pad: Adjustable bed pad features a quiet pump to alternately inflate and deflate individual bubble air cells; 4 LPM (liters per minute) pump produces consistent air flow and pressure over a 5 minute cycle time and is available fixed or variable
- 130 Air Cells: Medical mattress overlay has individual bubble air cells to provide therapeutic treatment of pressure ulcers for maximum comfort and effectiveness
- Pressure Sore Mattress Pad: Air pressure mattress for bed sores alternately inflates and deflates individual bubbles, which helps in the prevention, treatment, and management of pressure ulcers
- Durable: The alternating pressure mattress overlay is made of a heavy gauge, premium vinyl and has a weight capacity of 300 pounds
- Specifications: Measures 78 x 36 x 2.5 inches with a weight capacity of 300 lbs
| UPC | 822383135809 |
|---|---|
| Size | 78" x 36" (Variable Pressure Pump) |
| Color | Creme |
| Capacity | 136 Kilograms |
| Brand Name | Drive Medical |
| Item Depth | 36 inch |
| Model Name | AZ_58_14001e |
| Unit Count | 1 Count |
| Item Weight | 6.72 ounces |
| Manufacturer | Drive Medical |
| Model Number | 14001E |
| Fill Material | Air |
| Material Type | Vinyl |
| Cover Material | Vinyl |
| Item Thickness | 2.5 Inches |
| Item Dimensions | 12.5 x 8 x 7.25 inches |
| Number of Items | 1 |
| Product Benefits | Prevents and treats pressure ulcers with alternating pressure therapy |
| Construction Type | Vinyl pad with compressor pump and alternating bubble air cells |
| Additional Features | Adjustable |
| Included Components | Pad, Pump |
| Age Range Description | Adult |
| Water Resistance Level | Waterproof |
| Weight Capacity Maximum | 300 Pounds |
| Item Firmness Description | Extra Plush, Firm, Plush |
| Product Care Instructions | Wipe Clean with Damp Cloth |
| Specific Uses For Product | Bed, Sleeping Pad |
| Item Dimensions L x W x Thickness | 78"L x 36"W x 2.5"Th |
| Manufacturer Warranty Description | Limited lifetime warranty. |
| Global Trade Identification Number | 00822383135809 |
Correction of product information
If you notice any omissions or errors in the product information on this page, please use the correction request form below.
Correction Request Form